Pharma Outcomes That Matter and How Intelex Helps Deliver Them
February 20, 2026
8 minute read

In pharmaceutical manufacturing, the difference between a routine audit and a serious regulatory escalation often depends on the evidence you can provide. That evidence needs to clearly show that your data is consistent across all systems, sites, and regulatory responses.
When EHS and Quality operate on disparate platforms, audit risks and operational issues increase. Pharmaceutical manufacturers require integrated EHS and quality management systems that deliver five critical operational outcomes: inspection readiness, effective corrective actions, production continuity, proactive risk identification, and cross-site visibility.
These outcomes determine whether an organization maintains its license to operate or scrambles to remediate after the fact. In this article, we’ll explore why these outcomes matter and how Intelex delivers them.
Outcome 1: Always being inspection-ready with consistent answers
In pharma, inspection readiness shouldn’t be a quarterly sprint to organize spreadsheets and reconcile conflicting data. Ideally, it’s a continuous operational state.
When FDA or EMA inspectors walk into a facility (announced or not), the ability to produce consistent, defensible answers within hours determines whether an organization faces warnings, consent decrees, or smooth approvals.
The challenge isn’t a lack of data — most pharma companies are drowning in it. The problem is fragmentation. EHS tracks incidents in one system. Quality manages deviations in another (often validated and expensive) system. Operations uses manufacturing execution systems.

Safety, operations, and compliance data often live in disconnected systems, creating fragmented visibility and management challenges across the same organization
When an inspector asks about a chemical exposure event from three months ago and its connection to batch quality, teams scramble across disconnected platforms, manually piecing together timelines, and hoping nothing contradicts.
This inconsistency creates audit risk that goes beyond individual findings. Inspectors notice when the story changes depending on who’s answering. They question control systems, extend their stay, and turn what should have been a routine inspection into an enterprise crisis requiring executive attention.
How Intelex helps
Intelex provides a single source of truth across EHS, Quality, and Operations. When a contractor reports a chemical exposure in an aseptic filling suite, the system automatically links the incident to relevant batch records, quality investigations, and environmental monitoring data.
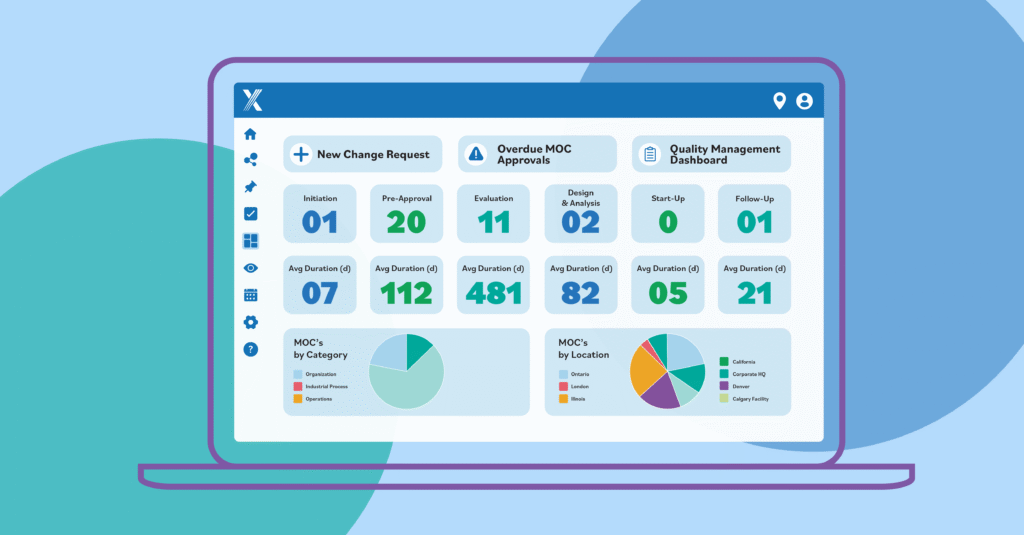
Intelex’s centralized dashboard displays key performance indicators across incident investigations, audits, observations, training, and more
No manual reconciliation needed — teams work from the same system. When inspectors ask questions, organizations answer confidently and consistently because the data already aligns, not because anyone rehearsed.
Inspection preparation shifts from weeks of frantic coordination to routine data extraction, reducing both the burden and the risk.
Outcome 2: Fewer repeat issues and provable CAPA effectiveness
Repeat findings carry a cost that extends beyond regulatory fines.
The same deviation across multiple batches, similar safety incidents at different sites, CAPAs that close on paper but resurface six months later — these patterns erode more than compliance scores. Organizations lose credibility with regulators, leadership loses faith in management systems, and teams waste resources fighting the same fires over and over.
Two problems drive this cycle: fragmentation and inadequate follow-through.
First, the fragmentation problem. Incidents and deviations with shared underlying issues get managed in isolation because Quality and EHS use separate processes and systems.
Quality investigates product deviations separately from EHS incident reviews. This separation obscures the true root cause, which often connects both domains, preventing organizations from addressing the actual issue.
Second, the follow-through problem. Even when organizations identify root causes correctly, CAPAs close when corrective actions are implemented, with no verification that the fix actually held or that similar issues are emerging elsewhere in the organization.
How Intelex helps
The Intelex platform feeds incidents and deviations with shared root causes into the same CAPA, regardless of which department initiated the investigation. When an ergonomic injury in packaging correlates with product damage trends, both issues connect to a unified corrective action plan.
Organizations get end-to-end traceability from issue identification through root cause analysis to effectiveness verification, building sustainability into the fix.
CAPA effectiveness gets proven through integrated data showing the problem hasn’t recurred across any connected system. This moves organizations from closing issues on paper to sustaining solutions in practice.
Outcome 3: Meeting compliance requirements without disrupting production
Pharmaceutical manufacturing operates under a fundamental tension: Compliance requirements are non-negotiable, yet production downtime costs millions per day.
This tension intensifies in controlled manufacturing environments, such as sterile filling suites, facilities handling cytotoxic compounds, and Process Safety Management facilities. Every audit, inspection protocol, and compliance verification potentially interrupts carefully controlled processes.
The traditional approach treats compliance and production as competing priorities. Audit preparation pulls operators off the floor for interviews. Environmental monitoring requires production pauses. Safety inspections delay batch releases.
To manage the tension, teams resort to last-minute workarounds that satisfy immediate compliance needs while creating longer-term risks through incomplete documentation or rushed implementations.
How Intelex helps
Intelex embeds EHS and Quality workflows directly into daily operations. This makes audit protocols part of standard operating procedures, not special events that require production shutdowns.
Operators complete safety observations on mobile devices without leaving the manufacturing floor. Environmental monitoring data flows automatically from equipment sensors. Standard operating procedures live in one central location that teams can access to complete tasks correctly, reducing errors and inconsistencies in the auditing process.
Production continues when auditors arrive. Because compliance data is already being generated as part of normal work, teams can maintain both regulatory requirements and manufacturing continuity simultaneously.
Outcome 4: Identifying and addressing risk earlier
Reactive risk management — i.e., responding after incidents occur — is the baseline in pharma. It’s required, auditable, and familiar. But reactive approaches are expensive, disruptive, and increasingly insufficient.
Using the reactive approach, organizations typically identify contamination risks, ergonomic hazards, and process safety gaps only after incidents have occurred. This approach carries both direct costs (production shutdowns, product holds, regulatory penalties) and indirect costs (reputational damage, inspector scrutiny, consent decree risk).
The barrier isn’t a lack of commitment to proactive approaches. Organizations remain stuck in reactive mode because they lack visibility across fragmented systems. When safety data, quality metrics, and operational performance exist in separate platforms, pattern recognition requires manual analysis that never happens because teams are too busy fighting fires.
Proactive risk identification represents both a competitive and operational advantage. Early pattern recognition prevents expensive incidents and creates competitive advantages through faster time-to-market, lower insurance premiums, and enhanced regulatory standing.
How Intelex helps
By connecting data across EHS, Quality, and Operations, Intelex surfaces correlations that would otherwise remain hidden in separate systems.
When near-miss incidents at one facility correlate with environmental excursions at another, the platform reveals these connections early enough for leadership intervention. When audit observations reveal emerging trends in a specific process area, teams can act before minor issues escalate into major findings.
This shift from reactive to proactive risk management delivers measurable advantages. Early pattern recognition prevents expensive incidents, accelerates time-to-market by avoiding regulatory delays, reduces insurance premiums through demonstrated risk control, and strengthens regulatory standing with agencies that value prevention over remediation.
The same transparency that streamlines audits also enables predictive risk management, moving organizations to the proactive side of the risk curve where prevention costs less than correction.
Outcome 5: Visibility across sites and teams with usable data
Global pharmaceutical operations generate massive volumes of EHS and Quality data, thousands of inspections, hundreds of incidents, and continuous environmental monitoring across dozens of facilities operating under different regulatory jurisdictions.
Most organizations cannot compare this data across sites without extensive manual analysis.
Site leaders know their local performance. Regional teams struggle to identify cross-facility patterns. Corporate leadership receives reports that are either too aggregated to be useful or too detailed to process.
When M&A activity adds new facilities with different systems and practices, the challenge multiplies. The opportunity cost is high: systemic risks go unrecognized, best practices don’t scale, and resource allocation decisions happen without complete information.
How Intelex helps
Intelex turns EHS and Quality data into clear, actionable insights. Organizations create a consistent structure across all sites while maintaining flexibility for local regulatory requirements.
Teams can drill down from enterprise-level dashboards to individual incident details, seeing both site-specific performance and systemic patterns.
Root cause analysis becomes truly comparative — teams see whether incidents at different facilities share underlying causes. Performance benchmarking happens automatically.
The flexible location structure eases the pains associated with mergers and acquisitions. Organizations can reorganize hierarchies, add new sites, and realign reporting relationships without disrupting ongoing operations or losing historical data.
This means acquired facilities integrate seamlessly into the existing organizational framework, maintaining their current structure during transition periods while gradually aligning to corporate standards.
When organizations make acquisitions, new sites integrate into the same system, accelerating the path to consistent, audit-ready operations across the entire enterprise.
Systems designed for the outcomes
Pharmaceutical organizations succeed or fail based on a small number of critical outcomes: passing inspections consistently, preventing repeat findings, maintaining production continuity, identifying risks before they escalate, and understanding performance across complex global operations.
These outcomes require integrated systems that connect EHS, Quality, and Operations into a unified platform. When data transparency, workflow automation, and cross-functional visibility work together, regulatory requirements shift from operational burden to strategic advantage.
The question isn’t whether an organization needs better outcomes. The question is whether current systems are designed to deliver them.
Ready to see what’s possible?
Use our demo builder to create your own personalized online experience. Choose from our pre-made demos or handpick features from our EHSQ solutions to make it your own.